New Diaphragm Hits U.S. Market for First Time in 50 Years
Caya, a new single-size diaphragm, gives women who can't or don't want to use hormonal methods a new birth control option.
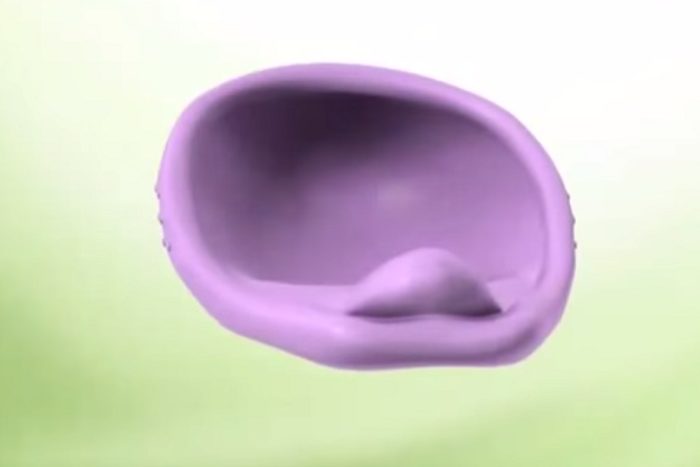
A newly designed diaphragm is hitting the U.S. market for the first time since the mid-1960s. Known as Caya, the new, single-size contraceptive device is comprised of a coiled nylon spring, a silicone cup, and a removal tab.
Caya is already widely available in Europe and was approved for sale by the U.S. Food and Drug Administration in September 2014. Its developers say Caya is easier to use than older models of diaphragms and believe that it’s a good option for women looking for non-hormonal methods of contraception.
The diaphragm was one of the original modern methods of contraception. Introduced in Europe in the 1880s, it was sometimes referred to as the Dutch Cap. It is a flexible dome-shaped cup that sits inside the vagina and covers the cervix, providing a barrier to sperm.
Women usually put a small amount of spermicidal jelly on the inside of the diaphragm for added protection, fold it in half, and insert it high into the vagina. It can be put in place hours before sex and used for multiple acts of intercourse over a 24-hour period.
Most diaphragms come in a variety of sizes and women need to have a pelvic exam where a health-care provider determines which size is best for them. If a woman gains or loses ten pounds, or has a baby, she is told to return to her provider to be resized.
The diaphragm is 94 percent effective if used perfectly, and 88 percent effective under typical use. That means that out of 100 couples who say this is their primary method of contraception, about 12 will become pregnant in the first year. In comparison, under typical use, the pill is considered 91 percent effective and the intrauterine device (IUD) is more than 99 percent effective.
Fewer and fewer women in the United States have turned to the diaphragm as birth control in recent years. In 1982, 17 percent of sexually active women had ever used a diaphragm, compared to 3.1 percent in 2010, according to the National Survey of Family Growth.
The developers of Caya hope that this new product might attract more users.
Caya was created through a collaboration between PATH (a Seattle-based global health nonprofit), CONRAD (a reproductive health product development organization operated through Eastern Virginia Medical School), the U.S. Agency for International Development (USAID), and other partners. The ten-year development process included feedback from test users. The final product includes suggestions from those who tested prototypes, such as a removal tab that makes the diaphragm easier to remove, especially for new users.
In the United States, Caya is being produced and marketed by HPSRx.
“In talking with family planning providers about this single-size diaphragm over the past few months, their eyes ‘light up’ when they hear that a new diaphragm is coming to market and no fitting exam is required,” Bob Patane, HPSRx’s founder, said in a statement. “Reducing barriers at the provider level could help bring this new method to a whole new generation of women who have not known that diaphragms are even an option.”
Studies have shown that Caya has similar efficacy rates to traditional diaphragms and that correct use was easy.
“In the recently published contraceptive effectiveness study, 76 percent of women were able to insert and correctly position the diaphragm simply using instructions. With coaching, 94 percent of women were able to insert, correctly position, and remove the diaphragm,” Gustavo Doncel, scientific and executive director of CONRAD, said in a press release. “This should make it easier to provide and use consistently.”
Most of the new contraceptive methods introduced in recent years—such as the contraceptive ring or the new versions of the IUD—contain hormones similar to those found in the birth control pill.
Women who can’t use hormonal methods because of pre-existing medical conditions and those who don’t want to use hormones have few options in the market. The introduction of Caya may appeal to them.

